
Kristen Conn
Assistant Professor, Department of Veterinary MicrobiologyResearch Area(s)
- Virology, virus-cell interactions, and molecular virology
Academic Credentials
- B.Sc. (Hon) Biochemistry, University of Saskatchewan
- Ph.D. Biochemistry, University of Alberta
Dr. Conn specializes in virology and virus-cell interactions. As a graduate student, she discovered that histone dynamics are dysregulated during herpes simplex virus infection and focused on characterizing this phenomenon throughout her Ph.D. During her postdoctoral training at the MRC-University of Glasgow Centre for Virus Research, she investigated the antiviral roles of small ubiquitin-like modifier (SUMO) regulatory proteins. Dr. Conn became a faculty member in the Veterinary Microbiology Department at the University of Saskatchewan in July 2018.
Research Interests
Constitutively expressed cellular proteins with antiviral activities make up the first line of intracellular defense against viral infection. These cellular proteins work together to restrict the replication and spread of viruses. In order to successfully replicate, viral proteins must consequently prevent or overcome these intracellular antiviral defenses. The individual mechanisms of the intracellular antiviral defense and how viruses counteract them are not yet fully known.
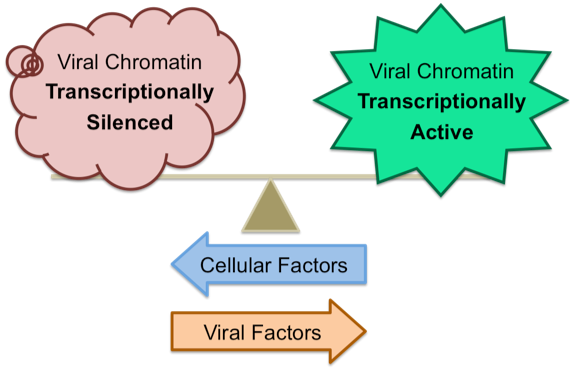
Our lab is interested in the roles that chromatin and chromatin regulatory proteins play in the intracellular antiviral defense. Transcriptionally silencing the infecting viral genomes would restrict viral protein expression and presumably weaken the virus’ ability to antagonize intracellular antiviral defenses. It is our model that cellular antiviral factors promote the transcriptional silencing of viral genomes while viral factors counteract cellular silencing attempts and promote the activation of viral transcription. The ability of the cellular or viral proteins to successfully silence or activate viral transcription, respectively, would ultimately determine the outcome of infection.
We use herpesviruses to investigate the chromatin regulation of viral gene expression and the roles that chromatin and chromatin regulatory proteins play in the intracellular antiviral response and how they are antagonized by viral proteins.
Publications
1) Alandijany T, Roberts A, Conn KL, Loney C, McFarlane S, Orr A and Boutell C. Distinct temporal roles for the promyelocytic leukaemia (PML) protein in the sequential regulation of intracellular host immunity to HSV-1 infection. PLoS Pathog. 2018; 14(1): e1006769. DOI:10.1371/journal.ppat.1006769.
2) Gibeault RL, Conn KL, Bildersheim MD, Hendzel MJ, and Schang LM. An essential viral transcription activator modulates chromatin dynamics. PLoS Pathog. 2016; 12(8): e1005842. DOI:10.1371/journal.ppat.1005842.
3) Brown JR*, Conn KL*, Wasson P, Charman M, Tong L, Grant K, McFarlane S, and Boutell C. The SUMO ligase protein inhibitor of activated STAT1 (PIAS1) is a constituent PML-NB protein that contributes to the intrinsic antiviral immune response to herpes simplex virus 1 (HSV-1). J. Virol. 2016; 90(13): 5939-52. Epub April 2016. *Equal Authorship
4) Conn KL*, Wasson P*, McFarlane S, Tong L, Brown JR, Grant KG, Domingues P, and Boutell C. Novel role for protein inhibitor of activated STAT 4 (PIAS4) in the restriction of herpes simplex virus 1 by the cellular intrinsic antiviral immune response. J. Virol. 2016; 90(9): 4807-26. Epub March 2016. *Equal Authorship
5) Tsai K, Chang L, Gibeault R, Conn K, Dheekollu J, Domsic J, Marmorstein R, Schang LM, and Lieberman PM. Viral reprogramming of the Daxx histone H3.3 chaperone during early Epstein-barr virus infection. J. Virol. 2014; 88(24): 14350-63. Epub October 2014.
6) Conn KL, Hendzel MJ, and Schang LM. The differential mobilization of histones H3.1 and H3.3 by herpes simplex virus 1 relates histone dynamics to the assembly of viral chromatin. PLoS Pathog. 2013; e1005575. DOI: 10.1371/journal.ppat.1005575.
7) Conn KL and Schang LM. Chromatin dynamics during lytic infection with herpes simplex virus 1. Viruses. 2013; 5(7): 1758-86.
8) Gombos RB, Brown JC, Teefy J, Gibeault RL, Conn KL, Schang LM, and Hemmings DG. Vascular dysfunction in young, mid-aged, and aged mice with latent cytomegalovirus infections. Am. J. Physiol. Heart Circ. Physiol. 2013; 304(2): H183-H194. Epub November 2012.
9) Conn KL, Hendzel MJ, and Schang LM. Core histones H2B and H4 are mobilized during infection with herpes simplex virus type 1. J. Virol. 2011; 85(24): 13234-52. Epub October 2011.
10) Conn KL, Hendzel MJ, and Schang LM. Linker histones are mobilized during infection with herpes simplex virus type 1. J. Virol. 2008; 82(17): 8629-46. Epub June 2008.

