Neospora caninum
Neospora caninum is a protozoan parasite, closely related and in several respects similar to Toxoplasma gondii, that is a major cause of abortion in cattle around the world and an occasional cause of neurological disease in young calves, dogs and other mammalian hosts.
Summary
Neospora caninum is a protozoan parasite, closely related to Toxoplasma gondii, that is a major cause of abortion in cattle around the world and an occasional cause of neurological disease in young calves, dogs and other mammalian hosts. In canid definitive hosts (dogs, wolves, coyotes), the parasite undergoes cycles of merogony and gametogony in the intestinal epithelium, resulting in the passage of unsporulated oocysts in feces. Oocysts sporulate in the environment. When an intermediate host (almost any mammal) ingests a sporulated oocyst, tachyzoites undergo rapid division and spread systemically, eventually forming tissue cysts containing bradyzoites in nervous tissue and skeletal muscle. The life cycle is completed when canids ingest tachyzoites or bradyzoites in the tissues, abortuses, or fetal membranes of intermediate hosts. In dogs and intermediate hosts, transmission can be vertical from dam to foetus, from new infections and/or recrudescence of a pre-existing Neospora infection, and can occur over multiple pregnancies.Neospora infection can be present in all, some, or none of the pups in litters from an infected dam, and disease can develop in all, some, or none of the infected pups. Many dogs infected with Neospora do not show disease. Clinical signs, which are more common in young dogs than in older animals, affect primarily the nervous system and skeletal muscles. In older dogs, a nodular, ulcerative dermatitis has been reported. Diagnosis in live animals is based primarily on serology and/or detection of the parasite or DNA in a biopsy, aspirate, CSF, or blood.
Female dogs who have previously had infected pups or are seropositive for Neospora should not be bred. Several drugs, particularly long-term clindamycin, have been used to alleviate the clinical signs of canine neosporosis, but do not clear the infection. To prevent exposure of dogs to Neospora, dogs should not be fed raw meat, especially from cattle and deer, and all mammalian tissues should be soldily cooked or frozen prior to feeding dogs. The parasite is not thought to be zoonotic.
Taxonomy
Phylum: Alveolata
Subphylum: Apicomplexa
Class: Coccidea
Order: Eimeriida
Family: Sarcocystidae
Neospora caninum is a relatively newly recognized apicomplexan protozoan related to the coccidians Toxoplasma, Sarcocystis, and Cystoisospora. It shares many biological features with Toxoplasma gondii. Another species within the genus, Neospora hughesi, occurs in horses. Neospora caninum was first described in 1988 as a cause of neurological disease in dogs, and then as a cause of abortion in cattle. Oocysts were first described from experimentally infected dogs in 1998.
Note: Our understanding of the taxonomy of parasites is constantly evolving. The taxonomy described in wcvmlearnaboutparasites is based on Deplazes et al. eds. Parasitology in Veterinary Medicine, Wageningen Academic Publishers, 2016.
Morphology
Unsporulated oocysts may be found in the feces of dogs and wild canids serving as definitive hosts for Neospora. Sporulated oocysts in the environment have two sporocysts, each containing 4 sporozoites. Although nothing is known of the life cycle stages in the enterocytes of the small intestine, presumably these take the form of meronts, microgamonts, and macrogamonts, similar to other coccidians.
In dogs as well as a wide range of intermediate hosts, intracellular tachyzoites can be found in any tissue, and thick-walled tissue cysts containing bradyzoites typicallyoccur in nervous tissue and skeletal muscles.
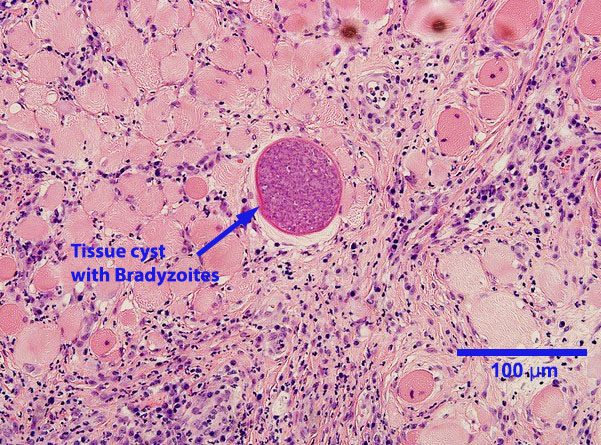
Tachyzoites occur singly or in clusters. Individual tachyzoites vary in shape from round to crescent-moon and measure up to approximately 7 μm by 5 μm. In cattle, tachyzoites can be found in many organs and tissues. Neospora tissue cysts containing bradyzoites in cattle are found in the brain and spinal cord, and possibly other locations. They measure up to approximately 100 μm in diameter, with a cyst wall 1 to 4 μm thick. Bradyzoites, which are found within the tissues cysts, are usually slender and measure up to approximately 7 μm by 1.5 μm.
Host range and geographic distribution
Neopora caninum has been identified in dogs and wild canids (dingos, wolves, coyotes, foxes) serving as definitive hosts in many countries around the world. Under natural conditions, the parasite utilizes carnivore definitive hosts and herbivore intermediate hosts. Many mammalian carnivores, including cats, can also serve as intermediate hosts.
The most important naturally infected intermediate hosts for N. caninum are thought to be cattle and white-tailed deer. In North America, serological or histological evidence of infection has also been found in sheep, goats, bison, black-tailed deer, caribou, moose, muskoxen, raccoons, cats, and polar bears, and elsewhere in the world in many other species. Currently in North America there are two transmission cycles for N. caninum: domestic – centred on dogs and cattle; and sylvatic – such as coyotes and white-tailed deer, wolves and caribou, etc. It is very likely that there is cross-over between domestic and sylvatic life cycles.
Life cycle
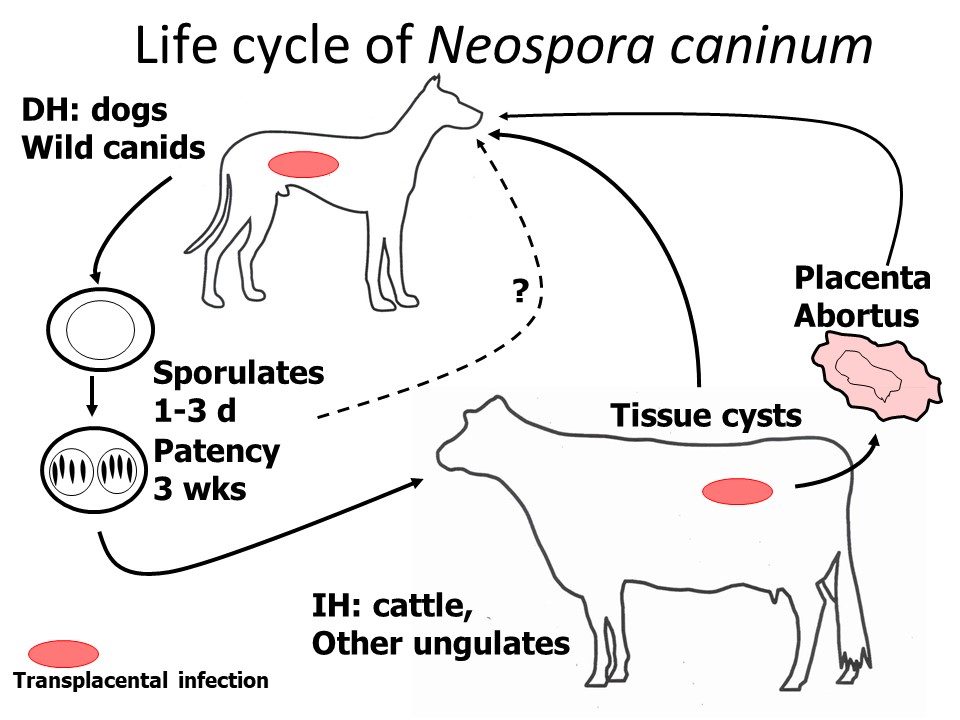
N. caninum alternates between a canid definitive host, in which the parasite produces oocysts in feces following development in the intestinal mucosa, and intermediate hosts, in which it forms extra-intestinal tissue cysts.
Dogs can also harbour the tissue stages of the parasite (tachyzoites, and tissue cysts containing bradyzoites) typical of intermediate hosts. As definitive hosts, dogs acquire the parasite by ingestion of tissues containing tachyzoites and/or bradyzoites from an infected intermediate host, or, possibly, by ingesting sporulated oocysts from the feces of another canid definitive host. Intermediate hosts become infected following ingestion of sporulated oocysts from the feces of an infected definitive host. Prenatal transmission is important in both definitive and intermediate hosts . .
Development of N. caninum in the definitive host that results in the production of oocystsis assumed to occur in the intestine (as with Toxoplasma in cats). The pre-patent period is 5-7 days. Oocysts are unsporulated when passed in the feces, and require a few days to sporulate and become infective. Each sporulated oocyst contains two sporocysts, each with four sporozoites. Oocyst production from experimentally infected dogs is often at low levels. Oocysts are shed for approximately three weeks, but may continue intermittently for several months.
In the intermediate hosts following ingestion of sporulated oocysts, sporozoites invade the intestinal wall and become tachyzoites, which divide asexually and disseminate throughout the body. After about 3 weeks, tissue cysts containing bradyzoites start to form.
Congenital infection (vertical transmission) occurs in both dogs and intermediate hosts when tachyzoites cross the placenta. This can result from new infections, or from recrudescence of a pre-existing Neospora infection. In dogs, only some of the pups born to infected dams become infected prenatally. An infected dam can pass the parasite to the foetuses during several successive pregnancies, but the risk of infection decreases with each pregnancy. It is also possible that colostrum and/or milk are important routes of transmission for pups.
Epidemiology
Pathology and clinical signs
Many dogs infected with Neospora caninum show no signs of disease. Clinical signs are seen most often in young dogs (from weaning to 1 year of age) that most probably acquired the parasite from their dams, either in utero or possibly through the colostrum and/or milk. The severity of disease seems to be greater in younger animals, especially those that acquire the infection prenatally. Disease is sometimes also seen in older animals, often in the form of skin ulcers. The signs most commonly observed in dogs of all ages affect the nervous system and skeletal musculature (polyradiculoneuritis/myositis) and usually begin with some form of incoordination or paresis of the hind limbs which may develop into rigid paralysis and ascend to involve the forelimbs. Other signs may include difficulty swallowing, megesophagus, muscle wasting, and incontinence. Neospora caninum has been recovered from many other organs and tissues of dogs (e.g., skin, lungs, and liver), and in some cases the lesions in these sites can lead to clinical signs. Unlike cattle, abortions due to Neospora have not been reported in dogs.
Diagnosis
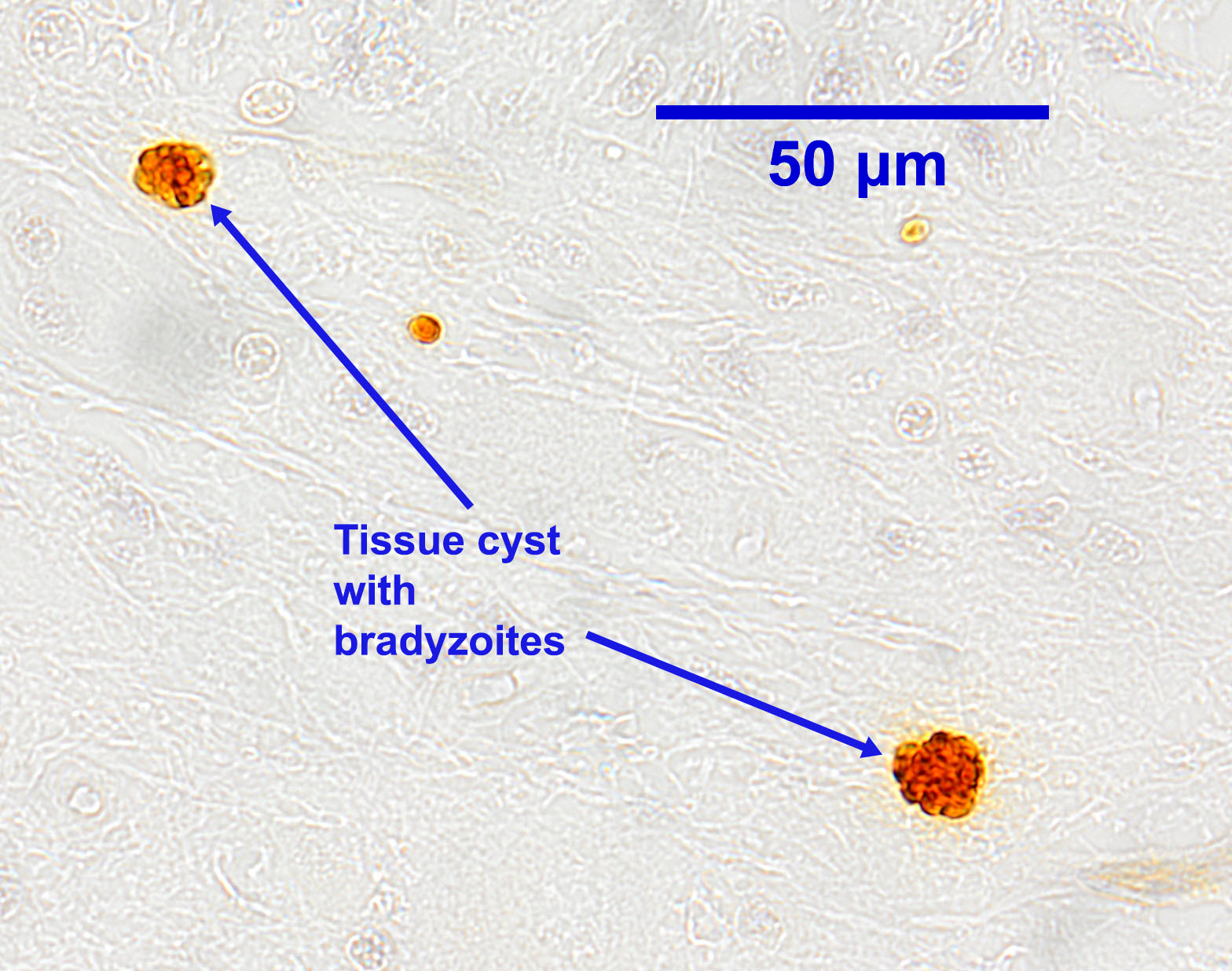
In live animals,history,clinical signs, and serology are very helpful in the diagnosis of neosporosis in dogs. Commercially available tests include a Neospora IFAT and a competitive ELISA. The IFAT is generally considered the gold standard, withgood sensitivity and specificity, and titres give an indication of the likelihood of Neospora being responsible for disease rather than simply exposure to the parasite. The test can also be used on CSF. Biopsies and tissue aspirates have also been used to diagnose Neospora in dogs, but immunohistochemistry or DNA-based techniques are needed to confirm the specific identification of coccidian tissue cysts. A recent case was successfully diagnosed using PCR on blood, skin, and CSF, combined with detection of tachyzoites in cutaneous aspirates.
Oocysts of N. caninum have been recovered only very rarely from naturally infected dogs, suggesting that patent infections in this host are rare and/or transient and/or produce only small numbers of oocysts. Like other coccidian oocysts, routine fecal flotation methods are appropriate for detection, although centrifugal methods are best. Unsporulated oocysts of N. caninum measure approximately 11µm by 11µm, smaller than Cystoisospora oocysts, which are much more common. Oocysts are similar in size to those of the non-pathogenic Hammondia heydorni in dogs, requiring DNA-based methods to differentiate. Tachyzoites and tissue cysts of Neospora in dogs can be tentatively diagnosed at post-mortem on the basis of typical pathological lesions, but as with biopsies and aspirates, immunohistochemistry or DNA-based techniques must be used to confirm the specific identification. Tachyzoites have been detected in many organs and tissues, but tissue cysts have been found only in nervous system tissues and, less commonly, skeletal muscles.
Treatment and control
Several drugs, most notably long-term clindamycin (+/- sulfonamides), have been used to treat clinical neosporosis in dogs. Sulfonamides and pyrimethamine may also be used. The aim of treatment is resolution of the clinical signs and/or slowing of clinical deterioration. For optimal results, therapy should be started as soon as possible after the diagnosis is established, and prognosis is better in older animals with more slowly progressing disease. Treatments do not inactivate tissue cysts and will not stop vertical transmission of Neospora from dam to offspring.
To prevent vertical transmission, no female dog that is seropositive for Neospora should be bred. In situations where Neospora is a particular concern (e.g., littermates of the dog are infected; the dam had previous litters with infected pups; or the parasite is present in cattle on the property) it might be worthwhile to serologically screen female dogs prior to breeding. To prevent horizontal transmission, dogs should not be fed raw meat, especially from cattle and deer. Meat and organs should be thoroughly cooked or frozen solid for 3 days prior to feeding to dogs. It is also important to minimize access by dogs to other sources of infection, for example aborted bovine foetuses and foetal membranes, as well as tissues from other potential intermediate hosts. Feces of dogs should be removed daily, before oocysts can sporulate. Finally, to prevent completion of the life cycle, dogs and wild canids should be prevented from fecally contaminating livestock feed and pasture whenever possible.
Public health significance
Despite the serological detection of human exposure to Neospora caninum in several regions of the world, to date there is no evidence for detection of the parasite, or its DNA, in people.
References
Decôme, M., Martin, E., Bau-Gaudreault, L., & O'Toole, E. (2019). Systemic disseminated Neospora caninuminfection with cutaneous lesions as the initial clinical presentation in a dog. Canadian Veterinary Journal 60(11), 1177–1181. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6805022
Lyon C (2010) Update on the diagnosis and management of Neospora caninum infections in dogs. Topics in Companion Animal Medicine 25: 170-175.
Dubey JP et al. (2009) Toxoplasmosis and other intestinal coccidial infections in dogs and cats. Veterinary Clinics of North America Small Animal Practice 39: 1009-1034.
Dubey JP et al. (2007). Epidemiology and Control of Neosporosis and Neospora caninum. Clinical Microbiology Reviews 20: 323-367.
Dubey JP (2003). Review of Neospora caninum and neosporosis in animals. Korean Journal of Parasitology 41: 1-16. (ejournal available on the web)
Dubey JP et al. (1996). A review of Neospora caninum and neosporosis. Veterinary Parasitology 67: 1-59.

